Cold Shipping: The Complete Guide to Cold Chain Logistics
Some shipments cannot afford to arrive warm. A pallet of craft beer, a shipment of cancer biologics, a consignment of fresh-cut flowers, each one has a narrow temperature window. Step outside it, even briefly, and the product is compromised. The business takes the hit.
Temperature -sensitive products typically move through some of the most demanding logistics environments in the modern supply chain. From fresh food and beverages to pharmaceuticals and life sciences products, these shipments require strict environmental control to remain safe and effective.
Even minor temperature deviations during transit can lead to product loss, compromised quality, or regulatory non-compliance. Frozen goods may thaw, pharmaceuticals may lose efficacy, and perishable food products may become unsafe for consumption.
This is why cold shipping has become a critical component of global logistics operations.
Cold shipping refers to the process of transporting temperature sensitive shipments within a controlled environment to maintain a required temperature range from origin to destination. It is a key function within broader cold chain logistics, ensuring that products remain within a specific temperature range throughout the supply chain.
For industries that depend on temperature controlled shipping, maintaining cold chain integrity is essential to ensure product quality, protect brand reputation, and deliver products safely to customers.
This guide breaks down how cold chain logistics works in practice. It covers packaging, refrigerants, cold storage, transportation, monitoring, and regulatory compliance. Whether you are shipping perishable food, beverages, pharmaceuticals, or agricultural products, the principles here apply across the board.
What Is Cold Shipping?
Cold shipping is the process of transporting temperature-sensitive shipments inside a controlled environment so that products arrive within a defined temperature range. It sits inside the broader discipline of cold chain logistics, which covers storage, processing, and distribution as well.
The focus of cold shipping is specifically the transit phase: the window between when a product leaves a temperature-controlled facility and when it arrives at its next destination. That window is where most cold chain failures happen.
Cold chain shipping relies on five interconnected elements:
Temperature-controlled packaging
Refrigerants such as gel packs, dry ice, or phase change materials
Refrigerated transport equipment
Cold storage warehouses at staging points
Real-time monitoring systems
The term cold chain integrity refers to keeping the temperature environment unbroken across every handoff. A single break, called a temperature excursion, can be enough to spoil a product, void a pharmaceutical batch, or trigger a regulatory non-compliance event.
Cold Chain Shipping and the Supply Chain
Cold shipping does not operate in isolation. It sits inside a larger supply chain system that connects production, storage, transportation, and final delivery. At every point in that system, there is a risk of temperature deviation.
A typical cold chain shipping flow looks like this:
Origin > Cold storage > Refrigerated transport > Distribution hub > Last-mile delivery
Each handoff is a vulnerability. A product stored correctly in a cold storage facility can still be compromised during loading if dock doors stay open too long. A well-packaged shipment can experience a temperature excursion if it sits on a warm tarmac waiting for an air freight connection.
This is why cold chain operations require coordination across every link: warehouses, carriers, last-mile providers, and monitoring systems all need to be aligned. And it is why choosing logistics partners with genuine cold chain expertise matters as much as choosing the right packaging.
A significant amount of perishable food is lost before it reaches consumers in markets with underdeveloped cold chain infrastructure. Even in mature markets, cold chain gaps drive significant product loss and food safety incidents.
Cold Chain Management: Four Systems That Work Together
Cold chain management is the operational and technological framework that keeps temperature-sensitive products safe across the supply chain. Effective cold chain management integrates four components.
1. Packaging Systems
The first line of defense. Packaging determines how long a product can survive outside a refrigerated environment. Insulated containers, the right refrigerant, and watertight liners all buy time when transit conditions are not ideal.
2. Storage Infrastructure
Cold storage warehouses maintain stable environments between transport legs. Products should never be staged in ambient conditions while awaiting the next pickup.
3. Transportation Networks
Refrigerated trucks handle regional and last-mile delivery. Refrigerated containers move large volumes internationally. Air cargo is used for urgent, high-value cold chain shipments. Each mode has different capabilities, cost structures, and cold chain risk profiles.
4. Monitoring Technology
IoT sensors track temperature and humidity throughout the journey. Automated alerts flag temperature deviations the moment they occur. Data logs support regulatory compliance documentation. Without monitoring, cold chain management is largely guesswork.
These four systems do not work well in isolation. Cold chain management is only as strong as its weakest link.
Cold Chain Packaging: Building the First Layer of Protection
Cold chain packaging is the most controllable variable in cold shipping. A well-designed packaging system can maintain the required temperature range through delays, ambient heat, and rough handling. A poorly designed one can fail within hours.
Insulation Materials
The outer container determines how long a shipment resists external temperature change. Common insulation materials include:
Expanded polystyrene (EPS) foam: Widely used, low cost, good performance for standard cold shipping
Polyurethane foam: Higher insulation performance; used for pharmaceuticals and longer transits
Vacuum insulated panels (VIPs): The highest-performing option; used for long-haul or temperature-critical shipments
Each material has a different R-value (thermal resistance). The higher the R-value, the longer the packaging holds its temperature without additional refrigerant.
Refrigerants
Packaging insulates. Refrigerants actively keep the temperature down. The right refrigerant depends on the required temperature range and transit duration.
Packaging Best Practices
These steps prevent the most common cold shipping packaging failures:
Pre-cool both the product and the insulated container before packing. Refrigerants should maintain temperature, not drive it down.
Use watertight plastic liners with a minimum thickness of 2 mil to contain melt and prevent leaks.
Eliminate void spaces with cushioning material. Empty air pockets warm up faster and create uneven temperature distribution.
Label outer packaging clearly: "Perishable," "Keep Refrigerated," or "Keep Frozen" as appropriate.
• Validate packaging performance for the specific transit duration before using it at scale.
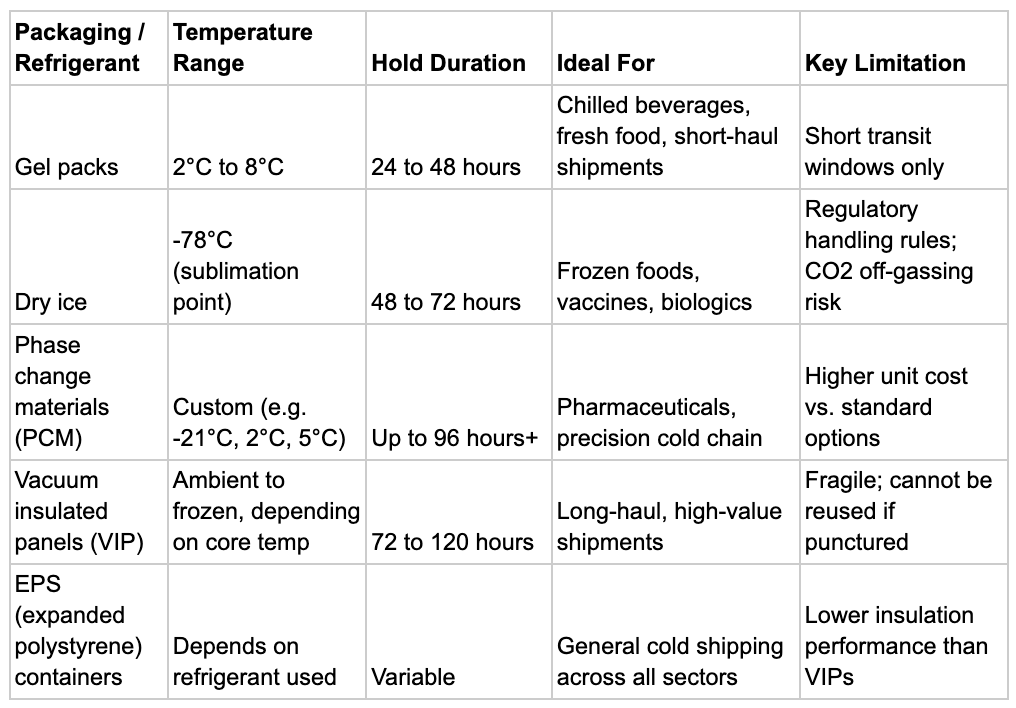
Refrigerant and Packaging Comparison
Choosing the right combination of insulation and refrigerant directly affects product safety, shipping costs, and transit flexibility. This table covers the most common options in cold chain shipping:
Dry Ice, Gel Packs, and Phase Change Materials
These three refrigerant types cover the vast majority of cold chain shipping needs. Understanding how each works prevents misapplication.
Gel Packs
Gel packs are water-based, flexible, and reusable. They are the standard refrigerant for chilled shipments in the 2 to 8 degree Celsius range. Used widely in food and beverage cold shipping, including wine, specialty beverages, and dairy products. They are safe, non-hazardous, and inexpensive. The limitation is duration: most gel pack configurations perform reliably for 24 to 48 hours.
Dry Ice
Dry ice is solid carbon dioxide. It sublimates directly from solid to gas without passing through a liquid phase, which means no mess and no water damage to products. It maintains temperatures down to approximately -78 degrees Celsius, making it essential for frozen pharmaceuticals, vaccines, and certain frozen food shipments.
Dry ice is a regulated hazardous material. Shipments must comply with IATA Dangerous Goods Regulations for air freight and DOT rules for ground transport. Carriers must be informed, and packaging must allow for CO2 off-gassing. These requirements add complexity and cost, but for frozen cold chain shipments, dry ice remains the most reliable option at scale.
Phase Change Materials (PCM)
Phase change materials are engineered to absorb or release heat at a specific, precise temperature. Unlike gel packs, which cool broadly, PCMs can be formulated to maintain a shipment at exactly 5 degrees Celsius, or exactly -21 degrees Celsius. This precision makes them the preferred refrigerant in pharmaceutical cold chain shipping where the temperature range is narrow and non-negotiable.
PCMs cost more per unit than gel packs or dry ice. For high-value or temperature-critical shipments, the cost is justified by the protection they provide.
Cold Storage and Temperature Controlled Warehousing
Cold storage warehouses are the staging points of the cold chain. Products enter them between production and transport, between transport legs, or ahead of last-mile delivery. What happens inside those facilities directly affects cold chain integrity.
Temperature controlled warehousing typically covers three zones:
Refrigerated storage: 2 to 8 degrees Celsius, used for chilled food, beverages, and some pharmaceuticals
Frozen storage: -10 to -25 degrees Celsius, used for frozen foods, ice cream, and certain biotech products
Ultra-low temperature storage: Below -60 degrees Celsius, used for mRNA vaccines and some advanced biologics
Poor warehousing practices are one of the most common causes of cold chain failure. Doors left open, overcrowded storage limiting airflow, inconsistent temperature zoning, and inadequate inventory rotation all create risk.
Best practices in cold storage include continuous temperature monitoring, strict first-in-first-out (FIFO) inventory rotation, documented receiving and dispatch procedures, and staff training on handling protocols. Facilities that hold food products in the US are also subject to FDA Food Safety Modernization Act (FSMA) requirements, including hazard analysis and preventive controls.
Monitoring Technology in Cold Chain Shipping
Real-time monitoring transforms cold chain operations. Without it, a temperature excursion might go undetected until a shipment arrives damaged. With it, logistics teams get an alert the moment a threshold is crossed.
Modern cold chain monitoring relies on IoT sensors embedded in packaging or transport vehicles. These sensors continuously record temperature and humidity data at defined intervals. Data is transmitted to cloud platforms where it is accessible in real time.
Automated alert systems notify logistics teams the moment a sensor detects a temperature breach. This allows immediate corrective action: rerouting a shipment, adding supplemental refrigerant, or flagging a batch for inspection before it reaches the customer.
Real-time telematics in refrigerated trucks add a second layer of visibility. Carriers can monitor trailer temperatures remotely, receive alerts for equipment malfunctions, and provide customers with documentation that the cold chain was maintained throughout transit.
Monitoring data also serves a critical compliance function. Pharmaceutical and food shipments often require documented proof that products stayed within the required temperature range at all times. Automated data logging makes this documentation straightforward rather than burdensome.
The International Air Transport Association (IATA) sets global temperature monitoring requirements for pharmaceutical air cargo shipments, including minimum data logger standards and documentation requirements.
Transit Planning for Cold Chain Operations
Transit time is one of the most controllable variables in cold shipping. Shorter transits reduce the total thermal stress on a shipment. Longer transits demand more from packaging, refrigerants, and monitoring systems.
Effective transit planning starts with route optimization. The fastest route is not always the most reliable. A shorter route with more handling points can introduce more temperature risk than a slightly longer direct route.
Timing matters too. Shipping at the start of the week reduces the risk of a shipment sitting in a carrier network over a weekend. Saturday and Sunday deliveries are often limited, and delays compound.
Contingency planning is part of professional cold chain management. What happens if a refrigerated truck breaks down? What is the procedure if a shipment is held at customs? These scenarios should be documented and rehearsed, not discovered during an incident.
Use direct routes where possible to minimize handling points
Ship early in the week, especially for multi-day transits
Build buffer time into transit windows to account for delays
Identify backup carriers or emergency cold storage options in key markets
Document contingency procedures and train logistics staff on them
Transportation in Cold Chain Shipping
Cold shipping relies on three primary transport modes. Each has distinct advantages, cost structures, and cold chain capabilities.
Refrigerated Trucks
The backbone of domestic cold chain shipping. Refrigerated trucks, sometimes called reefers, maintain active cooling throughout transit using diesel or electric refrigeration units. They are used for regional distribution, last-mile delivery, and full truckload or less-than-truckload cold chain shipments. In the US, the refrigerated trucking market is governed by the FDA's Sanitary Transportation of Human and Animal Food rule, which mandates temperature control requirements and documentation standards.
Air Cargo
Air freight is used for urgent, high-value, or perishable cold chain shipments that need to move quickly across long distances. Pharmaceuticals, fresh seafood, cut flowers, and specialty food products commonly move via air cold chain. The cost per kilogram is significantly higher than road or ocean freight, but transit times are dramatically shorter.
Refrigerated Containers (Reefer Containers)
For international cold chain shipping at scale, refrigerated intermodal containers are the standard solution. These containers maintain active temperature control during ocean freight and can be transferred directly to rail or truck without breaking the cold chain. They are used for large-volume food and beverage exports, pharmaceutical cold chain, and agricultural products.
Shipping Costs in Cold Chain Logistics
Cold chain shipping costs more than standard freight. The premium reflects the infrastructure, equipment, and expertise required to maintain temperature control across the supply chain.
Key cost drivers in cold shipping include:
Specialized packaging materials: Insulated containers, PCMs, dry ice, and VIPs all carry a higher per-unit cost than standard corrugated packaging
Active refrigeration: Refrigerated trucks, reefer containers, and climate-controlled air cargo add equipment and fuel costs
Monitoring technology: IoT sensors and real-time tracking platforms are priced into cold chain logistics services
Compliance and documentation: Meeting regulatory requirements for pharmaceutical or food cold chain shipping requires systems and processes that add overhead.
The cost of cold shipping must be weighed against the cost of cold chain failure. Product loss, regulatory penalties, customer refunds, and brand damage from a single temperature excursion incident can far exceed the savings from cutting corners on packaging or monitoring.
The most effective approach to controlling cold chain shipping costs is optimizing transit time, consolidating shipments, selecting the right packaging for the actual transit duration, and building relationships with carriers that have established cold chain infrastructure rather than patching together ad-hoc solutions.
Maintain Product Integrity and Food Safety
Maintaining product integrity is the core objective of cold shipping. Everything else, the packaging decisions, the monitoring systems, the carrier selection, exists to serve this goal.
For food and beverage products, the stakes include consumer health. Pathogenic bacteria multiply rapidly when temperature control fails. TheUS Department of Agriculture (USDA) identifies the temperature "danger zone" as between 40°F and 140°F (4°C to 60°C): the range in which bacterial growth accelerates. Cold shipping keeps food products below this threshold throughout transit.
Food safety regulations in the US tie directly to temperature control. FDA FSMA requires that carriers of refrigerated food maintain appropriate temperatures throughout transport and that shippers specify those temperatures in writing to carriers.
Beyond safety, product integrity covers quality. A bottle of wine that experiences a temperature spike during transit may not be unsafe, but its flavor profile is altered. A craft beer transported in ambient summer heat can develop off-flavors. A specialty spirit packaged without adequate cold chain protection in tropical climates can separate or cloud. These quality failures are not recalls, but they damage brand reputation and generate customer complaints.
For beverage brands shipping specialty, premium, or temperature-sensitive products, the cold chain is a brand investment, not just a logistics line item.
Cold Shipping in Life Sciences and Pharmaceuticals
No sector has higher cold chain stakes than life sciences. Vaccines, biologics, cell and gene therapies, and clinical trial samples require precise, validated temperature control. A deviation of a few degrees for a few hours can render a batch ineffective or unsafe.
The World Health Organization (WHO) estimates that temperature control failures account for a significant share of global vaccine waste each year. While estimates vary, cold chain failure remains one of the largest operational challenges in immunization programs worldwide.
Pharmaceutical cold chain shipping must comply with GDP (Good Distribution Practice) guidelines, which are published by organizations including the European Medicines Agency and the WHO. In the US, FDA regulations under 21 CFR Parts 210 and 211 govern storage and handling of drug products, including temperature requirements.
Pharmaceutical cold chain shipments require:
Validated packaging configurations tested for specific temperature ranges and transit durations
Calibrated temperature monitoring devices with documented data logs
Chain-of-custody documentation at every handoff
Qualified carriers with specialized pharmaceutical cold chain capabilities
Deviation reporting protocols when temperature excursions occur
The financial cost of cold chain failure in pharma is severe. Industry estimates suggest temperature excursions result in tens of billions of dollars in pharmaceutical losses globally each year.
Regulatory Compliance in Cold Chain Shipping
Regulatory compliance is not optional in cold chain shipping. For food, pharmaceutical, and life sciences products, maintaining accurate records and following defined protocols is a legal requirement.
In the US, key regulatory frameworks include:
FDA FSMA Sanitary Transportation Rule: Applies to food carriers, shippers, and loaders; mandates temperature control, vehicle cleanliness, and documentation
FDA 21 CFR Parts 210/211: Drug storage and handling requirements including temperature conditions
USDA Cold Storage and Grading Requirements: Apply to meat, poultry, and certain dairy products
IATA Dangerous Goods Regulations: Apply to dry ice and other hazardous refrigerants shipped by air
Across all regulated sectors, the compliance obligation comes down to three things: maintain the required temperature range, document that you maintained it, and have a written procedure for what happens when you do not.
Failure to comply carries real consequences: FDA warning letters, product recalls, import holds, and financial penalties. In the pharmaceutical cold chain, a single non-compliant shipment can trigger a batch rejection worth millions of dollars.
Cold Shipping for Beverages: A Special Consideration
Beverages sit in a category of their own within cold chain logistics. The range of cold shipping requirements across the beverage sector is wide.
Craft beer needs to remain cold to prevent skunking and off-flavor development. Premium wine, particularly temperature-sensitive varietals, should not be exposed to heat above 70°F during transit. Kombucha and probiotic beverages require refrigeration to maintain live cultures. Spirits and mixers may be less sensitive than perishables but are still affected by extreme heat, especially in export and tropical market contexts.
The volume and format of beverage shipments adds complexity. Cases of glass bottles are heavy, fragile, and dense. Kegs require different cold chain configurations than individual units. Bulk liquid shipments in reefer containers need precise temperature management throughout extended ocean transits.
Forsla specializes in the cold chain logistics of beverage products, managing the specific handling, temperature, and compliance requirements that come with shipping alcoholic and non-alcoholic drinks across markets. Understanding the unique cold chain needs of beverage brands, from small-batch craft producers to large-scale importers and exporters, is part of what makes beverage logistics a discipline of its own.
Common Risks in Cold Shipping and How to Prevent Them
Temperature Excursions
The most common cold chain failure. Often caused by equipment malfunction, delays in transit, or inadequate packaging for the actual transit duration. Prevention: validate packaging, monitor in real time, and build buffer time into transit windows.
Equipment Failure
A refrigeration unit failure mid-transit can compromise an entire load in hours. Prevention: partner with carriers that conduct pre-trip inspections, carry backup equipment, and have documented procedures for in-transit failures.
Improper Handling at Handoff Points
Loading docks, airport tarmacs, and distribution centers are where cold chain failures often happen. Prevention: train staff on cold chain handling protocols and minimize the time products spend in ambient conditions during transfers.
Documentation Gaps
In regulated sectors, missing temperature logs or incomplete chain-of-custody records can disqualify an otherwise intact shipment. Prevention: use automated data logging systems and audit documentation before shipments depart.
Industries That Depend on Cold Shipping
Cold chain logistics supports a wide range of sectors. Each has its own temperature requirements, regulatory landscape, and handling challenges.
Food and beverage: Fresh produce, dairy, meat, seafood, craft beverages, and specialty drinks
Pharmaceuticals and healthcare: Vaccines, biologics, clinical samples, and OTC medications that require refrigeration
Life sciences: Cell therapies, gene therapies, and laboratory specimens
Agriculture: Seeds, cut flowers, and perishable agricultural exports
Chemicals: Temperature-sensitive industrial chemicals and reagents
Floral industry: Cut flowers require refrigeration throughout transit to maintain freshness
Choosing the Right Cold Shipping Partner
The quality of a cold chain logistics partner determines whether products arrive intact and compliant. Carrier selection is not just a cost decision. It is a risk decision.
What to look for when evaluating a cold shipping provider:
Temperature-controlled fleet: Do they operate their own refrigerated trucks, or are they brokering to third-party carriers with variable cold chain standards?
Monitoring capability: Can they provide real-time temperature data and documented logs for each shipment?
Compliance expertise: Do they understand the regulatory requirements for the specific product categories you ship?
Cold storage network: Do they have access to cold storage warehouses at the staging points your shipments pass through?
Track record with your product type: Experience with pharmaceutical cold chain is different from experience with fresh produce or beverage logistics. Relevant sector experience matters.
What Good Cold Chain Logistics Actually Looks Like
Cold chain shipping done well is mostly invisible. Products arrive on temperature, documentation is clean, and nothing needs to be escalated. That outcome does not happen by accident.
It comes from choosing packaging validated for the actual transit duration. From partnering with carriers that operate real cold chain infrastructure, not just trucks with basic refrigeration. From monitoring systems that flag problems while there is still time to act. From transit planning that accounts for delays, not just best-case scenarios.
The global cold chain logistics market was valued at over$280 billion in 2023 and continues to grow as demand for temperature-sensitive food, pharmaceutical, and beverage products expands across new markets. The investment in cold chain infrastructure is not slowing down. Neither are the regulatory requirements or the consumer expectations around product quality and safety.
For brands and supply chain operators building cold shipping programs, the fundamentals have not changed. Know your product's temperature range. Validate your packaging. Choose partners with genuine cold chain capability. Monitor everything. Document it all.
Looking for a great partner for your cold shipping needs? Look no further than Forsla. Connect with us today to see how we make a difference to your logistics operations.
Frequently Asked Questions
What is cold chain shipping, and how does it differ from standard shipping?
Cold chain shipping is a specialized form of freight where temperature control is maintained across every transit leg, handoff, and storage point. Standard shipping does not account for temperature management. Cold chain shipments use insulated packaging, refrigerants, refrigerated transport, and real-time monitoring that conventional logistics does not require.
What causes a temperature excursion?
A temperature excursion occurs when a shipment moves outside its required temperature range. Common causes include equipment failure, transit delays, improper packaging, inadequate refrigerant for the transit duration, and mishandling at loading or delivery points.
What is the difference between gel packs and dry ice in cold chain packaging?
Gel packs maintain refrigerated temperatures (typically 2°C to 8°C) for 24 to 48 hours and are non-hazardous and reusable. Dry ice maintains frozen temperatures down to -78°C for 48 to 72 hours but is a regulated hazardous material that requires carrier notification and compliant packaging.
What are phase change materials in cold shipping?
Phase change materials (PCMs) are engineered refrigerants that absorb or release heat at a precise, predetermined temperature. They are used in pharmaceutical cold chain shipping where maintaining a narrow, exact temperature range is critical and standard gel packs are not precise enough.
How are cold storage warehouses used in cold chain logistics?
Cold storage warehouses hold temperature-sensitive products between transport legs. They operate in different temperature zones: refrigerated (2°C to 8°C), frozen (-10°C to -25°C), and ultra-low for specialized applications. Proper cold storage operations include continuous monitoring, FIFO inventory rotation, and controlled access to prevent temperature fluctuations.
Why do cold chain shipping costs run higher than standard freight?
Cold shipping costs more because it requires specialized packaging, refrigerants, active refrigeration equipment, real-time monitoring systems, and compliance documentation. These additions reflect the infrastructure required to protect temperature-sensitive products across the supply chain.
What regulations apply to cold chain shipping in the US?
Key US regulations include the FDA FSMA Sanitary Transportation Rule (food carriers and shippers), FDA 21 CFR Parts 210 and 211 (pharmaceutical storage and handling), and USDA requirements for meat, poultry, and dairy. Air freight shipments with dry ice also fall under IATA Dangerous Goods Regulations.
What temperature range is required for pharmaceutical cold chain shipments?
Most pharmaceutical products require refrigerated storage at 2°C to 8°C, but requirements vary by product. Some biologics and vaccines require frozen storage, and advanced therapies such as cell and gene therapies may require ultra-low temperatures below -60°C. Each pharmaceutical product has a validated temperature range specified in its regulatory approval documentation.
What does cold chain integrity mean?
Cold chain integrity refers to the uninterrupted maintenance of the required temperature environment across every stage of the cold chain, from production through storage, transit, and final delivery. A single break in this chain, at any handoff point, can compromise product safety, quality, or regulatory compliance.
What is the role of food safety in cold shipping?
Cold shipping is a frontline food safety control. Keeping perishable food products below 40°F (4°C) during transit prevents the bacterial growth that causes foodborne illness. US FDA FSMA regulations require that food shippers, carriers, and loaders maintain documented temperature control throughout the transport of refrigerated and frozen food products.